
OPTICAL IMAGING AND TRAPPING
Our group develops optical methods,
including optical tweezers, 3D high-resolution
bead tracking, and single molecule
fluorescence microscopy, to study the
dynamics and mechanics of single-molecules.
3D HIGH-RESOLUTION, FEEDBACK-STABILIZED OPTICAL TWEEZERS
Single-molecule techniques enable complex biomolecular interactions to be studied in a detailed way. In particular, molecular transitions can be observed directly rather than inferred from ensemble averages, and systems can be studied out of equilibrium and under force. Such investigations require the use of high-precision ultrasensitive force probes with nanometer length and piconewton force resolution.
We have developed a dual-mode optical tweezers system designed to explore both forward and reverse biomolecular transitions (e.g bond rupture & formation, protein unfolding & refolding). Our system incorporates a high-resolution 3D particle tracking technique based on reflection-interference imaging, as well as feedback systems which result in longtime stability of 1-2 nm. Our system is simple and accessible, yet powerful, using video microscopy with machine vision cameras for tracking.
In the two complimentary modes of operation, a functionalized glass probe bead is held by an optical trap near a reactive substrate, allowing single molecular tethers to bridge the two surfaces. The nanoscale motion of the probe bead reports molecular transitions such as bond rupture/formation and protein unfolding/refolding. The optical trap acts as a tunable spring, allowing precise and controllable application of force to the probe bead, and thus the molecular tether.
Horizontal Mode
for “far-from-equilibrium” studies
(e.g. bond rupture, protein unfolding)

High-resolution 1D tracking
-
Real-time video tracking and edge-detection
-
~2nm resolution@ 2500 samples/s

Active feedback for high-stability
-
Feedback loops for touch control, force application, and bead alignment
Vertical Mode
for “near-equilibrium” studies
(e.g. bond formation, protein refolding)

High-resolution 3D tracking
-
Reflection Interference Contrast Microscopy
-
~0.2 nm (z), ~1 nm (x-y) resolution @ 200 samples/s
-
Absolute distance measurement ideal for small (nm) distances

Active feedback for high-stability
-
Continuous autofocus system corrects for slow drift
-
Long-term stability: 1-2 nm in trap height, 10 fN in force
BEYOND THE FRAME RATE:
MEASURING HIGH-FREQUENCY FLUCTUATIONS WITH MOTION BLUR
Motion blur results from the finite exposure time (or shutter speed) of video cameras and other detection and acquisition systems. When properly understood, this effect is not a liability but instead provides valuable dynamical information. Below, we present two methods we have developed for using motion blur: one to calibrate force probes such as optical traps, and the second to measure the power spectral density (PSD) of any signal above the Nyquist frequency of an acquisition system.
Blur-corrected equipartition and power spectrum trap calibration
-
Simple and accurate method for determining the spring constant of a harmonically confined particle (e.g. optically trapped bead)
-
Corrects for motion-blur effect, and uses blur information to quantify the diffusion coefficient of the particle
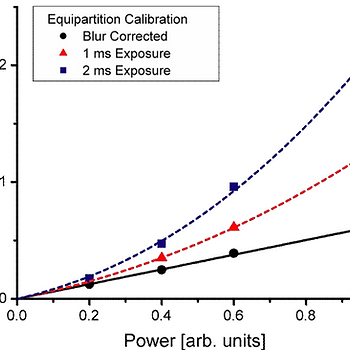

Experimental example with an optically trapped bead: Left: blur-corrected and uncorrected spring constant measurements as a function of laser power. Right: measured variance divided by true variance as a function of the dimensionless exposure time (exposure time divided by the trap relaxation time)
Blur-corrected equipartition and power spectrum trap calibration
-
By oscillating the signal source intensity, the power spectral density (PSD) can be determined at this frequency, even above the Nyquist limit
-
Overcomes acquisition rate and frequency response limitations of instruments, simply and inexpensively extending their effective dynamic range



IMSA Figures: Left: (a) Impulse responses and (b) frequency filters for DC and oscillating light Right: experimental example: IMSA measurement of power spectra for optically trapped beads
RELEVANT GROUP PUBLICATIONS
Beyond the frame rate: Measuring high-frequency fluctuations with light intensity modulation
WP Wong*^, K Halvorsen*. Optics Letters 34, 277-279 (2009). PDF
Power-spectral-density measurements of any sampled signal are typically restricted by both acquisition rate and frequency response limitations of instruments, which can be particularly prohibitive for video-based measurements. We have developed a new method called intensity modulation spectral analysis that circumvents these limitations, dramatically extending the effective detection bandwidth. We demonstrate this by video tracking an optically trapped microsphere while oscillating an LED illumination source. This approach allows us to quantify fluctuations of the microsphere at frequencies over 10 times higher than the Nyquist frequency, mimicking a significantly higher frame rate. Read more ›
Imaging biomolecular interactions by fast three-dimensional tracking of laser-confined carrier particles
We present a new technique to explore the dynamics of weak intramolecular interactions. It is based on the analysis of the 3D Brownian fluctuations of a laser-confined glass bead that is tethered to a flat surface by the biomolecule of interest. A continuous autofocusing mechanism allows us to maintain or adjust the height of the optical trap with nanometer accuracy over long periods of time. The resulting remarkably stable trapping potential adds a well-defined femto-to-piconewton force bias to the energy landscape of molecular configurations. A combination of optical interferometry and advanced pattern-tracking algorithms provides the 3D bead positions with nanometer spatial and >120 Hz temporal resolution. The analysis of accumulated 3D positions has allowed us not only to identify small single biomolecules but also to... Read more ›
The effect of integration time on fluctuation measurements: Calibrating an optical trap in the presence of motion blur
WP Wong*^, K Halvorsen*. Optics Express 14, 12517-12531 (2006). PDF
Dynamical instrument limitations, such as finite detection bandwidth, do not simply add statistical errors to fluctuation measurements, but can create significant systematic biases that affect the measurement of steady-state properties. Such effects must be considered when calibrating ultra-sensitive force probes by analyzing the observed Brownian fluctuations. In this article, we present a novel method for extracting the true spring constant and diffusion coefficient of a harmonically confined Brownian particle that extends the standard equipartition and power spectrum techniques to account for video-image motion blur. These results are confirmed both numerically with a Brownian dynamics simulation, and experimentally with laser optical tweezers. Read more ›
Exploring reaction pathways of single-molecule interactions through the manipulation and tracking of a potential-confined microsphere in three dimensions
WP Wong, V Heinrich, E Evans. Mat. Res. Soc. Symp. Proc. 790, P5.1.1-P5.1.12 (2004). PDF
We present a novel method to quantify subtle features of weak chemical transitions by analyzing the 3D Brownian fluctuations of a functionalized microsphere held near a reactive substrate. A weak optical-trapping potential is used to confine motion of the bead to a nanoscale domain, and to apply a controlled bias field to the interaction. Stochastic interruptions in the monitored bead dynamics report formation and release of single molecular bonds. In addition, variations in the motion of a bead linked to the substrate via a biomolecule (a protein or nucleic acid) signal conformational changes in the molecule, such as the folding/unfolding of protein domains or the unzipping of DNA. Read more ›





_jp.jpg)